Mass Of An Electron In Grams
News Co
Apr 05, 2025 · 6 min read
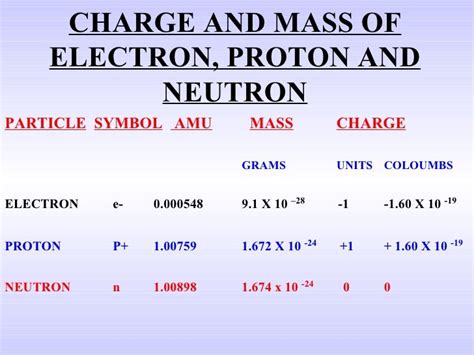
Table of Contents
The Mass of an Electron in Grams: A Deep Dive into Subatomic Physics
The electron, a fundamental particle of matter, is ubiquitous in the universe. Understanding its properties, particularly its mass, is crucial to comprehending the behavior of atoms, molecules, and ultimately, all matter. While the electron's mass is often expressed in atomic mass units (amu) or electronvolts (eV), its equivalent in grams provides a tangible connection to our everyday understanding of weight and scale. This article delves into the intricacies of determining the electron's mass in grams, explores the methods used for its precise measurement, and discusses its significance in various scientific fields.
Understanding the Electron's Mass: From Amu to Grams
The electron's mass is incredibly small, highlighting the immense scale at which subatomic particles operate. Its most commonly cited value is approximately 9.109 × 10<sup>-31</sup> kilograms. To convert this to grams, we simply multiply by 1000, yielding 9.109 × 10<sup>-28</sup> grams. This tiny number underscores the challenge in directly weighing an electron. The techniques used to determine this mass rely on indirect measurements and clever experimental design.
The Significance of Precise Measurement
The precise determination of the electron's mass isn't just an academic exercise. It plays a pivotal role in various scientific domains:
-
Particle Physics: Precise mass measurements are crucial for testing fundamental theories like the Standard Model of particle physics. Discrepancies between theoretical predictions and experimental results can point towards new physics beyond the Standard Model.
-
Atomic Physics: The electron's mass is fundamental to calculating the properties of atoms and molecules, including their size, energy levels, and chemical reactivity. Accurate mass values are essential for accurate simulations and predictions.
-
Astrophysics: Understanding the mass of fundamental particles is crucial for modeling stars, galaxies, and the universe's evolution. Mass-energy equivalence (E=mc²) directly links mass to energy, affecting processes like stellar nucleosynthesis and black hole formation.
-
Materials Science: Electron mass influences the electrical conductivity, magnetic properties, and other characteristics of materials. Precise measurements contribute to developing new materials with desired functionalities.
Methods for Determining Electron Mass: A Historical Perspective
The determination of the electron's mass wasn't a single event but a gradual refinement through several experimental techniques:
1. Millikan's Oil Drop Experiment (Charge-to-Mass Ratio)
Robert Millikan's groundbreaking oil drop experiment in 1909 didn't directly measure the electron's mass, but it provided a crucial piece of the puzzle: the charge-to-mass ratio (e/m). By observing the motion of charged oil droplets under the influence of electric and gravitational fields, Millikan determined the charge of an electron (e). Combining this with pre-existing measurements of e/m (obtained from studying cathode rays), the electron's mass (m) could be calculated. This was a monumental achievement, providing a relatively accurate estimate of the electron's mass.
2. Spectroscopy and Atomic Transitions
Spectroscopic techniques, analyzing the light emitted or absorbed by atoms, provide another avenue to determine the electron's mass indirectly. The energy levels of electrons within an atom are directly related to the electron's mass and the atomic nucleus's properties. By meticulously analyzing the spectral lines, scientists can infer the electron's mass through sophisticated calculations. The high precision of modern spectroscopic methods has contributed to increasingly accurate mass determinations.
3. g-2 Experiments (Anomalous Magnetic Moment)
Electrons possess an intrinsic magnetic moment, a property related to their spin. The precise measurement of this moment (g-factor) provides another means to determine the electron's mass. The "g-2" experiments focus on measuring the tiny difference between the theoretical value of g and the experimentally measured value, often called the anomalous magnetic moment. This difference is highly sensitive to subtle effects, including the electron's mass and interactions with other particles. The precision of g-2 experiments continues to improve, resulting in ever more refined mass determinations.
Challenges and Uncertainties in Mass Measurement
Despite significant advancements, challenges remain in achieving an absolutely precise value for the electron's mass:
-
Quantum Uncertainty: The inherent uncertainty principle of quantum mechanics limits the simultaneous precision of measuring position and momentum. This limitation affects the accuracy of experiments relying on particle tracking and detection.
-
Systematic Errors: Experimental setups are susceptible to systematic errors, such as imperfections in the instruments, environmental influences, and uncertainties in the calibration of measurement equipment. Careful error analysis and rigorous experimental design are crucial to minimizing such errors.
-
Higher-Order Corrections: Theoretical calculations of the electron's properties require incorporating higher-order quantum electrodynamic (QED) corrections. These corrections become increasingly complex and computationally challenging, potentially affecting the accuracy of indirect mass determinations.
The Electron's Mass in Various Units and its Interrelation
While the gram is a familiar unit, the electron's mass is often expressed in other units better suited for subatomic calculations:
-
Atomic Mass Units (amu): One amu is defined as 1/12th the mass of a carbon-12 atom. The electron's mass is approximately 0.00054858 amu. This unit is convenient for comparing the masses of atoms and subatomic particles.
-
Electronvolts (eV): The electronvolt is a unit of energy, but through Einstein's mass-energy equivalence (E=mc²), it can also be used to represent mass. The electron's mass is approximately 0.511 MeV/c², where MeV represents mega-electronvolts and c is the speed of light. This unit is widely used in particle physics.
The interrelation between these units highlights the versatility of scientific measurements and the importance of choosing the most appropriate unit for a particular context.
Beyond the Mass: Other Properties of the Electron
Understanding the electron's mass is crucial, but it's just one piece of the puzzle. Other essential properties include:
-
Charge: The electron carries a negative elementary charge, which is the fundamental unit of electric charge.
-
Spin: Electrons have an intrinsic angular momentum called spin, quantized in units of ħ/2 (reduced Planck constant). This spin is responsible for the electron's magnetic moment.
-
Wave-Particle Duality: Electrons exhibit both wave-like and particle-like behavior, a fundamental concept in quantum mechanics.
-
Lepton Family: The electron belongs to the lepton family of fundamental particles, which also includes muons and tau particles.
Conclusion: The Ongoing Quest for Precision
The determination of the electron's mass in grams, while seemingly a simple conversion, represents a significant achievement in scientific history. The methods employed showcase the ingenuity of experimental physicists and the power of theoretical frameworks like quantum mechanics and quantum electrodynamics. While the current value is highly precise, the quest for even greater accuracy continues, driven by the need to refine our understanding of fundamental physics and its implications across diverse scientific disciplines. The pursuit of a more accurate measurement of the electron's mass will undoubtedly lead to further breakthroughs and a deeper comprehension of the universe at its most fundamental level. The ongoing refinement of measurement techniques and theoretical models ensures that our understanding of this tiny, yet immensely significant particle, continues to evolve. The journey towards a more precise understanding of the electron's mass is a testament to the ongoing scientific endeavor, highlighting the boundless curiosity and persistent pursuit of knowledge that define the scientific spirit.
Latest Posts
Related Post
Thank you for visiting our website which covers about Mass Of An Electron In Grams . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.